No edit summary |
No edit summary |
||
| Line 17: | Line 17: | ||
I use red cabbage to make pH indicator. The pH solution from 2 to 13. | I use red cabbage to make pH indicator. The pH solution from 2 to 13. | ||
[[File:WechatIMG440.jpeg|400px]] | |||
1. pH=2 100ml distilled water + 1ml HCL | 1. pH=2 100ml distilled water + 1ml HCL | ||
[[File:WechatIMG418.jpeg|400px]] | |||
2. pH=4 100ml distilled water + 0.1g apple acid | 2. pH=4 100ml distilled water + 0.1g apple acid | ||
[[File:WechatIMG438.jpeg|400px]] | |||
3. pH=5.5 100ml distilled water + 2g H3BO3(Boric acid) | 3. pH=5.5 100ml distilled water + 2g H3BO3(Boric acid) | ||
[[File:WechatIMG433.jpeg|400px]] | |||
4. pH=7 100ml distilled water | 4. pH=7 100ml distilled water | ||
[[File:WechatIMG429.jpeg|400px]] | |||
5. pH=8 100ml distilled water + 0.5g NaHCO3 | 5. pH=8 100ml distilled water + 0.5g NaHCO3 | ||
[[File:WechatIMG436.jpeg|400px]] | |||
6. pH=11 700ml distilled water + 1ml NaOH | 6. pH=11 700ml distilled water + 1ml NaOH | ||
[[File:WechatIMG427.jpeg|400px]] | |||
7. pH=12 250ml distilled water + 1ml NaOH | 7. pH=12 250ml distilled water + 1ml NaOH | ||
[[File:WechatIMG424.jpeg|400px]] | |||
8. pH=13 100ml distilled water + 1ml NaOH | 8. pH=13 100ml distilled water + 1ml NaOH | ||
[[File:WechatIMG420.jpeg|400px]][[File:WechatIMG430.jpeg|400px]] | |||
Revision as of 16:14, 10 January 2018
References
I use red cabbage to make pH indicator. The pH solution from 2 to 13.
 1. pH=2 100ml distilled water + 1ml HCL
1. pH=2 100ml distilled water + 1ml HCL
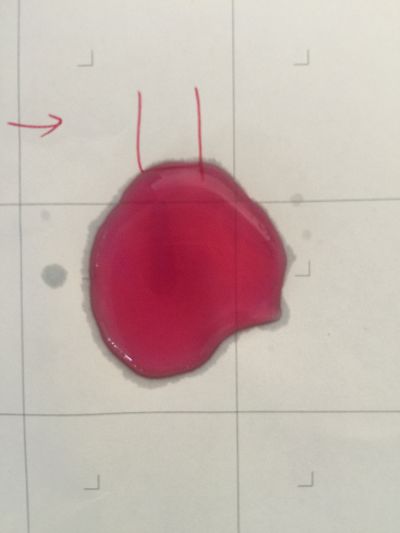 2. pH=4 100ml distilled water + 0.1g apple acid
2. pH=4 100ml distilled water + 0.1g apple acid
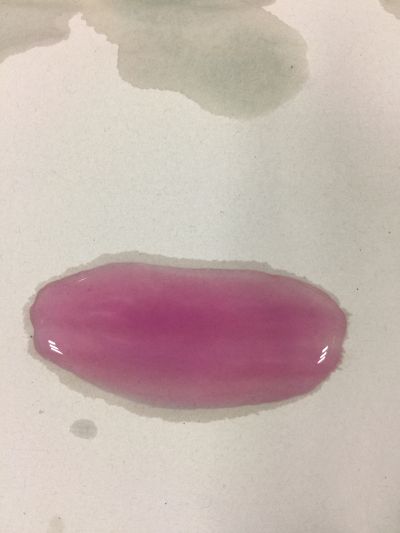 3. pH=5.5 100ml distilled water + 2g H3BO3(Boric acid)
3. pH=5.5 100ml distilled water + 2g H3BO3(Boric acid)
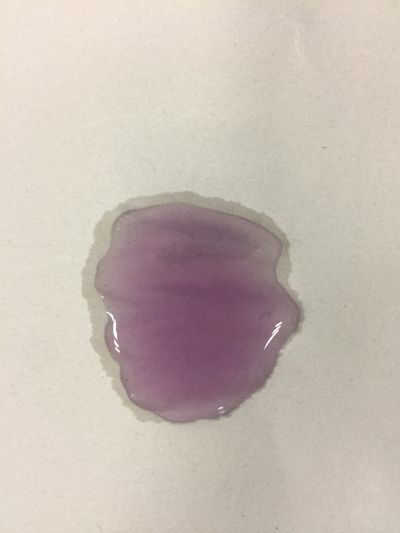 4. pH=7 100ml distilled water
4. pH=7 100ml distilled water
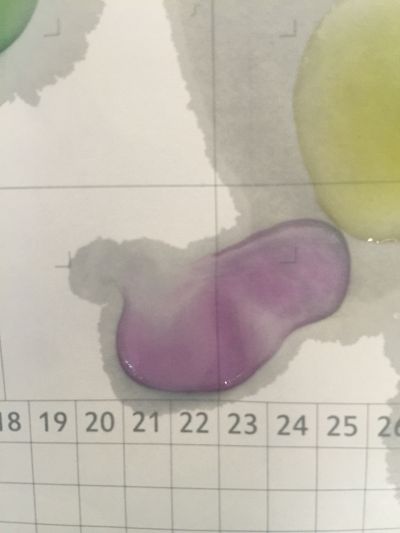 5. pH=8 100ml distilled water + 0.5g NaHCO3
5. pH=8 100ml distilled water + 0.5g NaHCO3
 6. pH=11 700ml distilled water + 1ml NaOH
6. pH=11 700ml distilled water + 1ml NaOH
 7. pH=12 250ml distilled water + 1ml NaOH
7. pH=12 250ml distilled water + 1ml NaOH
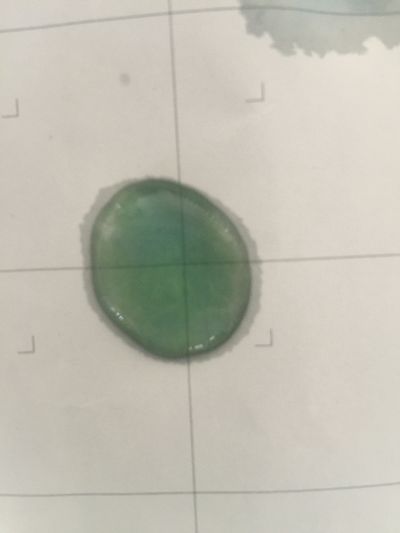 8. pH=13 100ml distilled water + 1ml NaOH
8. pH=13 100ml distilled water + 1ml NaOH
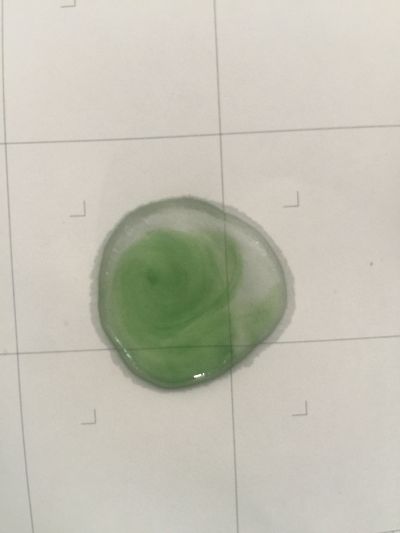

Next step:
1. Rate of colour change
2. Reversibility of colour change






